Understanding the Application of Fluid Conditioning: The Effect of Contamination
Water: In hydraulic fluids, excess water can degrade the fluid and the system in numerous ways, even though water may be present in small quantities. In general, water content up to 700 ppm (parts per million) may be tolerated. However, some tests and types of fluid indicate that levels as low as 140 ppm can significantly reduce pump and fluid life under certain circumstances. Water can cause serious degradation in system performance and life because of its effects on the fluid. Effects could include changes in viscosity and lubricity, increase in oxidation rate, precipitation of additives, and combination with chemicals to form acids. It is important to prevent water entry into the system and to be prepared to remove any that might find its way in. Water typically exists in three forms in hydraulic oils – free water, emulsified water, and water in solution.
Free water is water which can readily separate from the oil. This water will usually settle to the bottom of the reservoir and should be removed by periodic draining. Water can enter the system as free water during external cleaning, rain, sprinkler system operations, defective heat exchangers, and so forth. It can enter the system as water vapor from the atmosphere through a vented reservoir.
Emulsified water is the result of the mixing action of the pump on water-contaminated fluid or from high turbulence or violent agitation at points in components or fluid conveyors. These actions cause the water droplets to reduce in size so that they will not readily separate by settling or centrifuging. Some additives, including detergents, polar rust inhibitors and EP (extreme pressure) additives, can increase emulsification. Emulsified water in fluid imparts a milky appearance to the fluid. Severe emulsification (high water content) may give the oil a gelatinous texture.
Water in solution is dependent on the type of oil, base stock, additives, and temperature. The amount of water in solution typically ranges from 60 to 300 ppm (0.006 to 0.03%) at 100°F to 500 to 1,000 ppm (0.05 to 0.1 %) at 180°F. This water is not visible when in solution, but appears as a cloud in the oil as temperature is lowered to the critical temperature that begins to force the water out of solution.
Air and Gas: Air affecting hydraulic systems can be classified as dissolved, entrained, foam, and free. All hydraulic fluids contain dissolved air. This is air that is in solution; that is, the air molecules occupy the spaces (interstices) between the molecules of the liquid. The amount of air present is measured as a percentage of the liquid volume and varies with pressure. At sea level, the amount of dissolved air may be as high as 10%. One liter of hydraulic fluid could contain 100 mm of air. A minor drop in pressure will cause air to be drawn out of solution and become entrained.
Entrained air consists of bubbles that are less than 1 mm in diameter. A drop in pressure is one cause. It can occur locally during turbulent flow, when fluid returns to the reservoir, or when there is low inlet pressure to a pump. It can also be caused by hydraulic fluid entering the reservoir above the fluid level as the liquid stream drags air with it into the reservoir (see Figure 1).
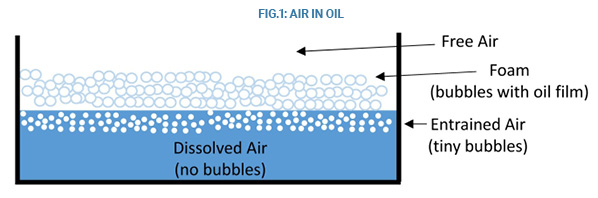
Foam is a condition where large numbers of air bubbles rise to the surface and gather together surrounded by an oil film.
Free air is the air above the fluid level in a reservoir, or pockets of air that are trapped somewhere in the circuit. This is the air that can be drawn into the hydraulic fluid and become entrained or produce foaming.
Air in the system can be the cause of a number of problems. Dissolved air can come out of solution as the liquid circulates through the system. The resultant air bubbles reduce the bulk modulus of the hydraulic fluid, leading to sponginess and erratic behavior of actuators. Air reduces the lubricating film between the moving parts in the system. Air bubbles will suddenly collapse under high pressure, causing locally high temperatures that can damage equipment. If foaming occurs, it can spill out into the environment, wasting fluid and making a mess.
At approximately 70°F (21°C), a vacuum of 5 in. Hg will pull the entrained air out of the oil.
Particulate matter: different sized particles affect clearances in different ways. If the particle is larger than a clearance or orifice, it may not enter the clearance and· cause interference, but it could block the opening. This could present a serious problem in the case of small damping or control orifices in components. Particles that are close to the size of the clearance may get caught between clearances and cause abrasive damage or jamming of the component. Very small particles, typically smaller than 4μ, are called silt. These particles usually pass through most clearances, but in high fluid velocity, turbulence or impingement on surfaces when flow direction changes, they may erode metal surfaces or metering control edges. This can reduce efficiency and create heat by increasing internal leakage. It can also affect response, control and reliability. This type of erosive damage generates additional wear particles that further aggravate contamination damage and failures.
The effect of fluid temperature: Hydraulic fluids are typically not considered high temperature fluids. Their useful life, as well as maintenance of viscosity, chemistry and the other attributes, is based upon continuous operation below a critical temperature. This critical temperature is 150°F (66°C). Every 18°F increment (10°C) higher than 150°F effectively doubles the oxidation rate of the hydraulic fluid (petroleum base) thus cutting its useful life in half. For example, running a system at a consistent 176°F (80°C) would reduce the useful life of the fluid by 75%. Most fluid manufacturers specify optimum ranges of temperature for their products, typically from 90°F to 120°F (32°C to 49°C) or 100°F to 130°F (38°C to 54°C) even though many fluids are operated in excess of these temperature ranges. The effects of temperature on the fluid are the same, regardless of the source of heat.
As the fluid heats up, it gets thinner. This increases internal leakage which in turn heats the fluid more. With most water based fluids, heat will cause evaporation of the water, change the ratio of water to base, and will increase viscosity. On the other hand, when temperatures are too low, the fluid thickens, increasing the energy required to move it through the system. This can cause pump cavitation and/or sluggish behavior of actuators. Water-based fluids will freeze and shut down the whole system if the temperature is low enough.
Thermal shock can occur when fluid that has been warmed is directed to an actuator that is very cold, as in hydraulically driven ski lift equipment. The dissimilar metals within a hydraulic motor, for example, may expand at different rates and, because of close tolerances, could lock up.
Viscosity: Properties that affect the performance of a hydraulic fluid include viscosity, specific gravity, viscosity index, pour point, neutralization number, flash point, fire point, auto ignition temperature, anti-wear properties, resistance to oxidation and rust, anti-foaming, and detergent dispersing properties. Of these factors, viscosity is the most important operational property.
The molecular structure of any fluid causes it to resist flow. This resistance is the viscosity. The higher the viscosity, the greater is the resistance to flow. Viscosity is measured either in Saybolt universal seconds (SUS), where a specific volume of fluid is timed as it runs down a calibrated tube at a controlled temperature, or in Centistokes, where a ball is dropped into a tube of fluid and timed as it descends. Hydraulic fluid is formulated to utilize this resistance to flow as a means of lubrication for the closely fitted parts that make up hydraulic components. The fluid adheres to surfaces, creating the thin film of lubricant necessary to allow the parts to slip by each other with maximum ease and minimal wear.
Viscosity is directly related to temperature. As the fluid warms up due to ambient conditions and inefficiencies, it becomes less viscous, making it easier to flow but reducing its ability to lubricate. Where there is a wide variation in the operating temperature of the fluid, it becomes necessary to use a multi-viscosity fluid that is less viscous at low temperatures and more viscous at higher temperatures. The rate of viscosity change due to temperature is called the viscosity index (VI).
There is no universal recommendation for the viscosity of hydraulic fluid. The requirements of the equipment manufacturer should be known and followed.
Energy tip: Energy is required to overcome the resistance to flow. This energy is a parasitic loss that detracts from the efficiency of a system. It is important to choose the fluid with the optimum viscosity for the application.
Cleanliness: Viscosity is the most important functional property of hydraulic fluid but cleanliness is the most important operational property. The wrong viscosity can cause premature wear or excessive energy loss but contamination, even in properly viscous fluid, can quickly destroy components. For this reason, target cleanliness levels have been established for various classes of components. When these cleanliness levels are met and the optimum hydraulic fluid is used, long and efficient life can be expected.
Test Your Skills
1. Water typically exists in three forms in hydraulic oils:
a. Ice crystals, gas, and condensation.
b. Free water, emulsified water, and water in solution.
c. Entrained water, milky water, and steam.
d. Cloudy oil, milky oil, and foam.
e. Liquid, gas, and solid.
2. Air affecting hydraulic systems can be classified as:
a. Dissolved.
b. Entrained.
c. Foam.
d. Free.
e. All of the above.
3. What are the most important operational and functional properties of a hydraulic fluid?
a. Viscosity and cleanliness.
b. Flash point and environmental impact.
c. Specific gravity and specific heat.
d. Pour point and vapor point.
e. Neutralization number and centistoke.
What are the correct solutions?
1.b, 2. e, 3. a







